Draft guidance on default values and uncertainty factors to be used by the EFSA Scientific Committee, Scientific Panels and Units in the absence of actual measured data
On this page
Skip the menu of subheadings on this page.This is a paper for discussion. This does not represent the views of the Committee and should not be cited.
Background
1. The European Food Safety Authority (EFSA) has updated its 2012 guidance on selected default values to be used by the EFSA Scientific Committee (SC), Scientific Panels and Units in the absence of actual measured data. Comments are being sought on the draft guidance. The closing date for comment is the 14th May 2026. The key features of the draft guidance are set out in the paper below.
2. Members are asked to send in any additional comments to the Secretariat by Friday 8th May citing the line and section number where possible. A shared document is also available in the Members area folder.
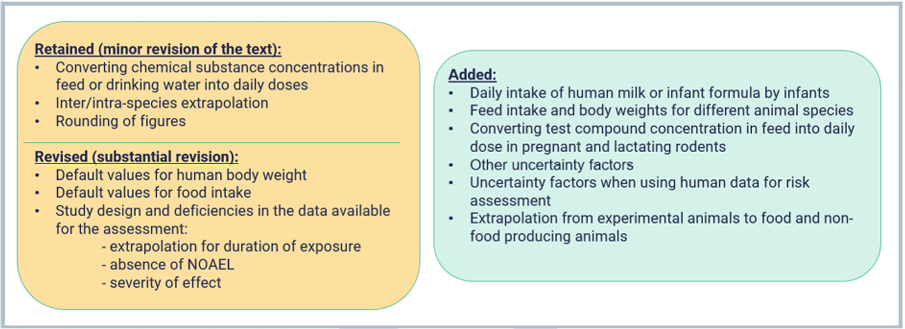
3. Box 1 (page12) of the draft guidance summarises the proposed changes to the 2012 guidance.
Default values
4. Default values (DVs) are representative or typical values for body weights of humans or animal species, food and feed intake, or factors used to convert the concentration of a chemical substance in feed or drinking water into daily doses in experimental animal studies. They are not intended to be conservative.
Body weights
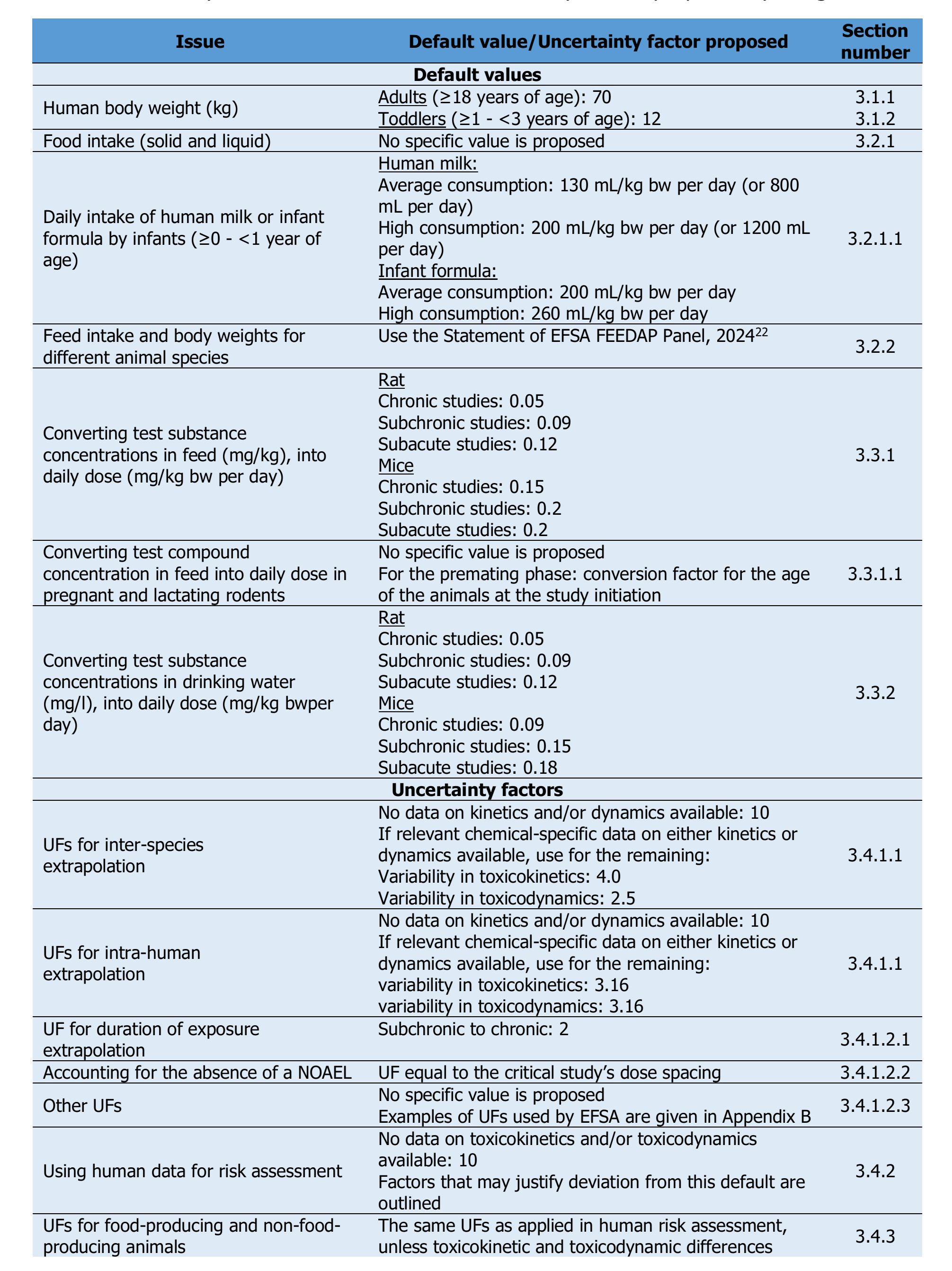
5. The draft guidance recommends that default values of 12 and 70 kg are used for toddlers of ≥ 1 to < 3 years, and adults respectively. This recommendation took into account the most recent version of the EFSA Comprehensive European Food Consumption Database but are the same values as recommended in the 2012 guidance (EFSA, 2012).
6. The draft SC guidance did not propose DVs for other specific population subgroups (e.g. pregnant women) and recommended that if values were required for the assessment of specific (age/sex) groups, the respective mean or relevant percentile values given in the guidance should be used. Alternatively, the EFSA Comprehensive European Food Consumption Database could be used to extract specific values.
7. The Food Standards Agency (FSA) use specific values from the National Diet and Nutrition Survey and where this not possible, a default of 70 kg, for an adult. The SC guidance notes that using a dietary exposure assessment for both toddlers and adults in risk assessments would encompass dietary exposures of older children and adolescents, which are likely to be intermediate between those of adults and toddlers. This is consistent with the approach taken by FSA in incident risk assessments.
Consumption
8. The draft guidance discusses default values for intakes of solid food and liquids in humans. It was noted that empirical data were available from the comprehensive database and that default values were no longer needed. However, default values were recommended for the consumption of human milk (mean and high level consumption of 130 and 200 ml/kg bw/day respectively) and infant formula (mean and high level consumption of 200 and 260 ml/kg bw/day respectively for exclusively formula fed infants).
9. The standard values of feed intake, concentration of the test substance in feed and default body weight of different animal species are used to estimate dietary exposure in animals. This approach is used in the risk assessments for food and non-food-producing animals. A recent statement from FEEDAP (2024) on dietary exposure from feed aimed to standardise these values across EFSA for along with food and water intakes for experimental animals.
Conversion from feed and water concentrations to dose
10. For conversion of the concentration of chemicals in feed or drinking water to dose (mg/kg bw) a number of factors are recommended in the draft guidance. These are based on WHO EHC 240 (IPCS, 2009) and the 2012 analysis of the applicability of these remains valid. It was noted that exposure would be higher in the first week of a study. While these values would largely apply to reproductive toxicity studies, OECD compliant teratogenicity studies were usually conducted using gavage and data would be available.
11. For drinking water, default conversion factors are recommended based on data from National Toxicology Program studies.
12. For risk assessment, the FSA currently uses the conversion factors recommended in EHC 240.
Uncertainty factors
13. Uncertainty factors (UFs) tend to be conservative. UFs are used to account for uncertainties in the assessment such regarding inter/intra-species extrapolation, extrapolation for duration of exposure or for deficiencies in the database.
Inter- and intra-species extrapolation
14. In the absence of chemical-specific data on kinetics and/or dynamics, the draft guidance recommends using the overall UF of 100 (10 for inter-species variability x 10 for intra-human variability). However, it states that the following default sub-factors should be considered:
- for inter-species variability in toxicokinetics: 4.0.
- for inter-species variability in toxicodynamics: 2.5.
- for intra-human variability in toxicokinetics: 3.16.
- for intra-human variability in toxicodynamics: 3.16.
15. It is further recommended that where available and relevant, chemical-specific data on kinetics and/or dynamics should be used to modify the overall UF of 100.
Limited database
16. The guidance recommends that where there are deficiencies in the available database, the assessment of whether additional data can be obtained to enhance the robustness of the dataset should be prioritised, rather than applying an additional UF. However, when additional data cannot be obtained, the application of an additional UF to account for deficiencies in the database should be considered on a case-by-case basis and justified. It was noted that a generic UF was not appropriate, as it will depend on the nature and extent of the available dataset.
Extrapolation for duration of exposure
17. The draft guidance noted the UFs recommended by the European Chemicals Agency (ECHA) (2012) of:
- subacute to subchronic: 3.
- subchronic to chronic: 2.
- subacute to chronic: 6.
18. The SC guidance recommends the use of a UF of 2 for extrapolation from sub-chronic study to chronic study duration, provided that measured parameters are relevant for chronic toxicity. However, there was less confidence in the effects observed in sub-chronic or chronic studies being apparent in studies of a shorter duration. It was noted that in cases where there is a need to use a subacute study as the basis for the risk assessment, the UFs recommended by ECHA could be considered, providing this study is sufficiently reliable and if all relevant endpoints covered in standard (sub)chronic toxicity studies have been investigated.
Accounting for the absence of a No-Observed-Adverse-Effect-Level (NOAEL)
19. The draft guidance states that the benchmark dose (BMD) approach should be used where possible. This can be used in the absence of a NOAEL without the need for an additional UF.
20. If the lower bound of the BMD or a NOAEL cannot be identified than the LOAEL may have to be used in the evaluation if the study is of appropriate quality. The magnitude of the effect at the LOAEL and the shape and steepness of the dose–response relationship should be taken into account when considering an appropriate UF to apply.
Other uncertainty factors
21. The draft SC guidance does not recommend the use of a default UF to account for the specific toxicological properties of the hazard, such as the severity of the critical effect or the potential of the substance to accumulate in the body. However, it is stated that in specific situations, the application of an additional UF should be determined through expert judgment and be justified on a case-by-case basis.
Using human data for risk assessment:
22. The use of human data for risk assessment means that there is no need for a UF to extrapolate from animals to humans. In the absence of data on variability in toxicokinetics and toxicodynamics among humans, the draft SC guidance recommends applying a UF of 10 to account for inter-individual differences.
Use of uncertainty factors for food-producing and non-food-producing animals:
23. The draft guidance states that where there are no species-specific data available, studies on experimental animals could be considered for hazard identification and characterisation for food-producing and non-food producing animals. The use of the same UFs as applied in human risk assessment are also recommended on a case-by-case basis, when assessing the risk for food-producing or non-food producing animals of chemicals than feed additives.
Rounding figures when deriving HBGVs
24. The draft guidance states that “communicating an estimated figure (e.g. an HBGV) with an excessive number of significant figures may convey a spurious idea of precision, masking the assumptions made and the UFs that were used to establish the HBGV”. The SC therefore recommends that values, such as health-based guidance values, should be rounded to a single significant figure if the impact of rounding is less than 10%, and to two significant figures if the impact of rounding to one significant figure exceeds 10%. It is noted that rounding should be done as late as possible in the assessment process.
Summary and recommendations
25. The draft guidance also makes a number of general recommendations:
- The SC recommends the evaluation of the scientific basis for the UF of 100 for inter and intra-species extrapolation and its sub-factors by an international expert group.
- Risk assessors should consider replacing the UFs by Chemical-Specific Adjustment Factors (CSAFs) to reduce the uncertainty in the risk assessment (e.g. when the metabolic pathway is known to be highly polymorphic).
- The SC recommends that where DVs defined in legislation are not compatible with the scientific basis, such legislation should be reconsidered and aligned with the current state of knowledge.
- The SC recommends checking the compatibility of the growth curves for humans included in toxicokinetic models with the data in the Comprehensive Database.
- The SC recommends that the use of probabilistic approaches should be investigated.
- The SC recommends that the proposed values and their possible impact for risk assessment should be discussed between relevant European bodies and international organisations.
26. Table 7 is taken from the draft guidance document and sets out the proposed DVs and uncertainty factors.
Questions for the Committee
27. Members are asked if they have any comments on the recommendations on the proposed use of:
a) Default values.
b) Uncertainty factors.
Secretariat
March 2026
References
ECHA 2012. European Chemicals Agency (ECHA). Guidance on information requirements and chemical safety assessment. Chapter R.8: Characterisation of dose[concentration]-response for human health. Guidance for implementation of REACH Guidance on information requirements and chemical safety assessment
EFSA FEEDAP 2024 Animal dietary exposure in the risk assessment of contaminants in feed Animal dietary exposure in the risk assessment of contaminants in feed - - 2024 - EFSA Journal - Wiley Online Library
EFSA (2012) EFSA Scientific Committee; Guidance on selected default values to be used by the EFSA Scientific Committee, Scientific Panels and Units in the absence of actual measured data. EFSA Journal 2012;10(3):2579, 32 pp. https://doi.org/10.2903/j.efsa.2012.2579
International Programme on Chemical Safety (IPCS), 2009. Environmental Health Criteria 70, Food additives and contaminants in food, principles for the safety assessment of food additives and contaminants in food. Geneva, Switzerland, World Health Organization, International Programme on Chemical Safety.
Annex A to TOX/2026/13
Link to draft guidance and excel spreadsheet containing details of the analysis: